Prediction and Prevention of Falls using an Implantable Cardiac Monitor



Origins

Each year, one in every three adults aged 65 and older will experience a fall. Falls are an evolving frailty state and are the most common reason for older adults to attend emergency departments and for admission to long term institutional care. TILDA has shown that almost 40% of older adults reported at least one fall during a four year period and almost 50% had 'fear of falling', an independent risk factor for falls and loss of independence. If preventive measures are not taken in the immediate future, the numbers of injuries caused by falls is projected to be 100% higher in the year 2030, with the healthcare associated costs projected to double by the year 2050. New mechanisms for monitoring early risk factors for falls are needed to advance prevention and management of these conditions.
The Study
This project is co-funded by EIT Health. The FFallS Predictor Clinical Investigation is a research study that aims to investigate the value of embedding a triaxial accelerometer within an implantable cardiac monitoring device (The Reveal LINQ™) to predict unexplained falls. 'Unexplained' or 'non accidental' falls are falls which are not clearly due to a slip or a trip. Previous research shows that a high number of these falls may be due to changes in heart rhythm and there may also be other associated changes such as overall activity levels.
The Falls and Syncope Unit (FASU) at St James's Hospital treats patients experiencing non-accidental falls, and in cases where a cardiovascular aetiology is suspected but not confirmed an implantable cardiac monitor (ICM) is indicated. The Reveal LINQ™ is an ICM system with the ability to monitor heart rate and rhythm activity and is pre-programmed to detect abnormalities. There is scope to further develop implantable devices such as the Reveal LINQ™ and to fully explore their capabilities to monitor additional physiological parameters related to falls.
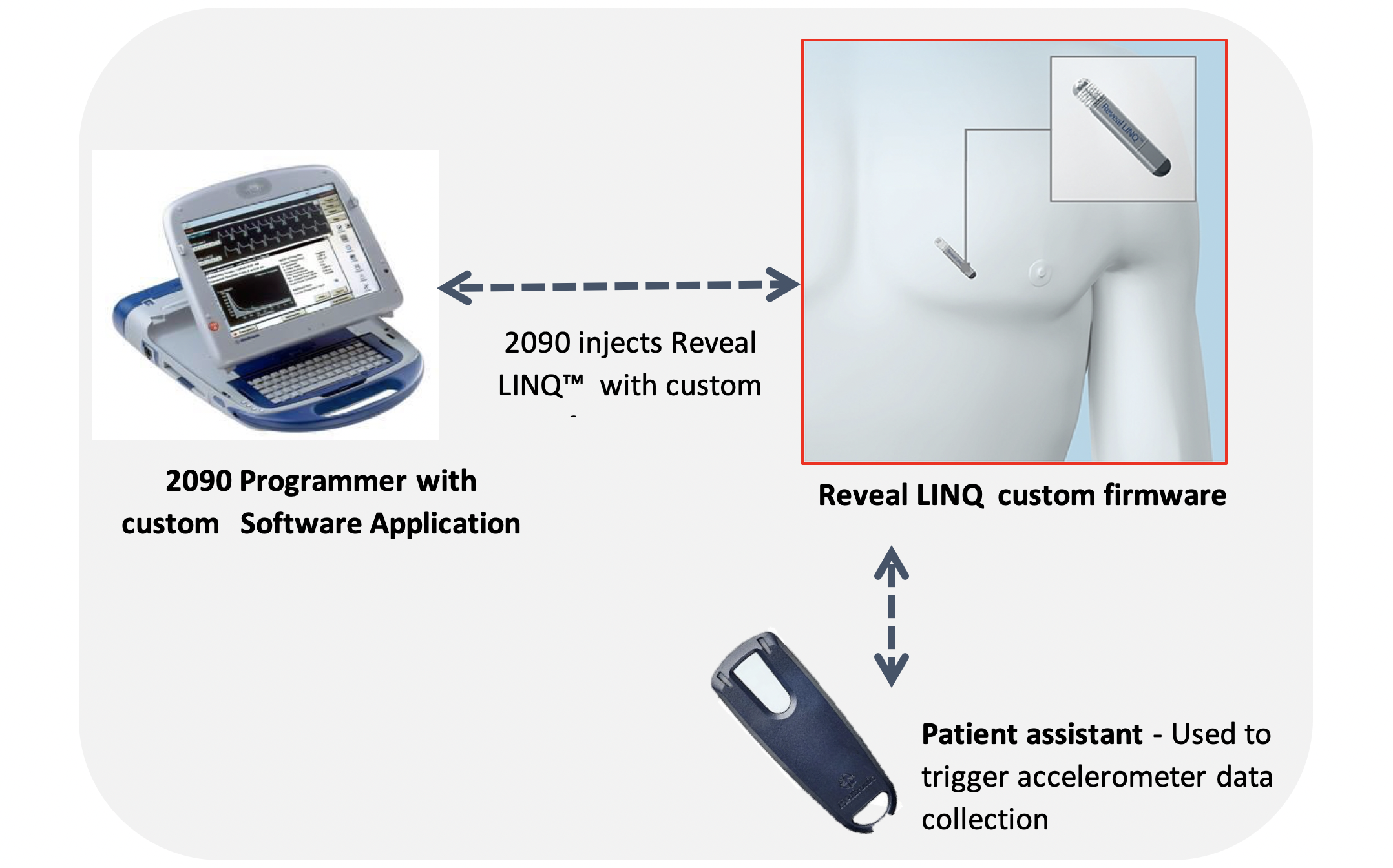
Medtronic in collaboration with the PI Prof Kenny have developed a custom firmware for the Reveal LINQ™ to collect tri-axial accelerometer data, which provides information on gait, posture and activity. The additional investigational data along with the standard cardiac parameters that are monitored may be useful in predicting or identifying physiological changes before a fall.

Objectives
• Evaluate the capability of the Reveal LINQ™ Falls Prediction Research System to identify early changes in physiological parameters as risk predictors of falls.
• To build on established research of the cardiac parameters of the Reveal LINQ ™ to identify heart rate and rhythm disturbances in fallers and to evaluate their role in predicting a fall.
• Develop a clinical risk stratification algorithm for management, treatment and prevention of frailty and falls.
Design
This prospective, single centre, pilot feasibility study recruited 30 participants (from recurrent non-accidental fallers referred to FASU) aged 50 years or older with a history of unexplained falls. Participants had a Reveal LINQ™ implanted and modified with the Falls Prediction (FP) firmware and completed a comprehensive baseline assessment, including physical examination, frailty assessment, gait assessment, ECG, and details of experienced falls in the past. These assessments were repeated every 3 months over the follow up period of 12 months. Details from falls were captured by participants on fall diaries, as well as by the clinical team through regular phone calls. In addition, the follow up period included a daily gait assessment, weekly manual transmission to the Carelink™ System and an additional 6-minute walking assessment during the scheduled in-clinic follow up visits. For the daily walk assessments participants were required to manually initiate collection of accelerometer waveform data using the use the Patient Assistant tool. Patients were asked to walk for 1-minute post activation to ensure 30 seconds of data collection.
Sample description
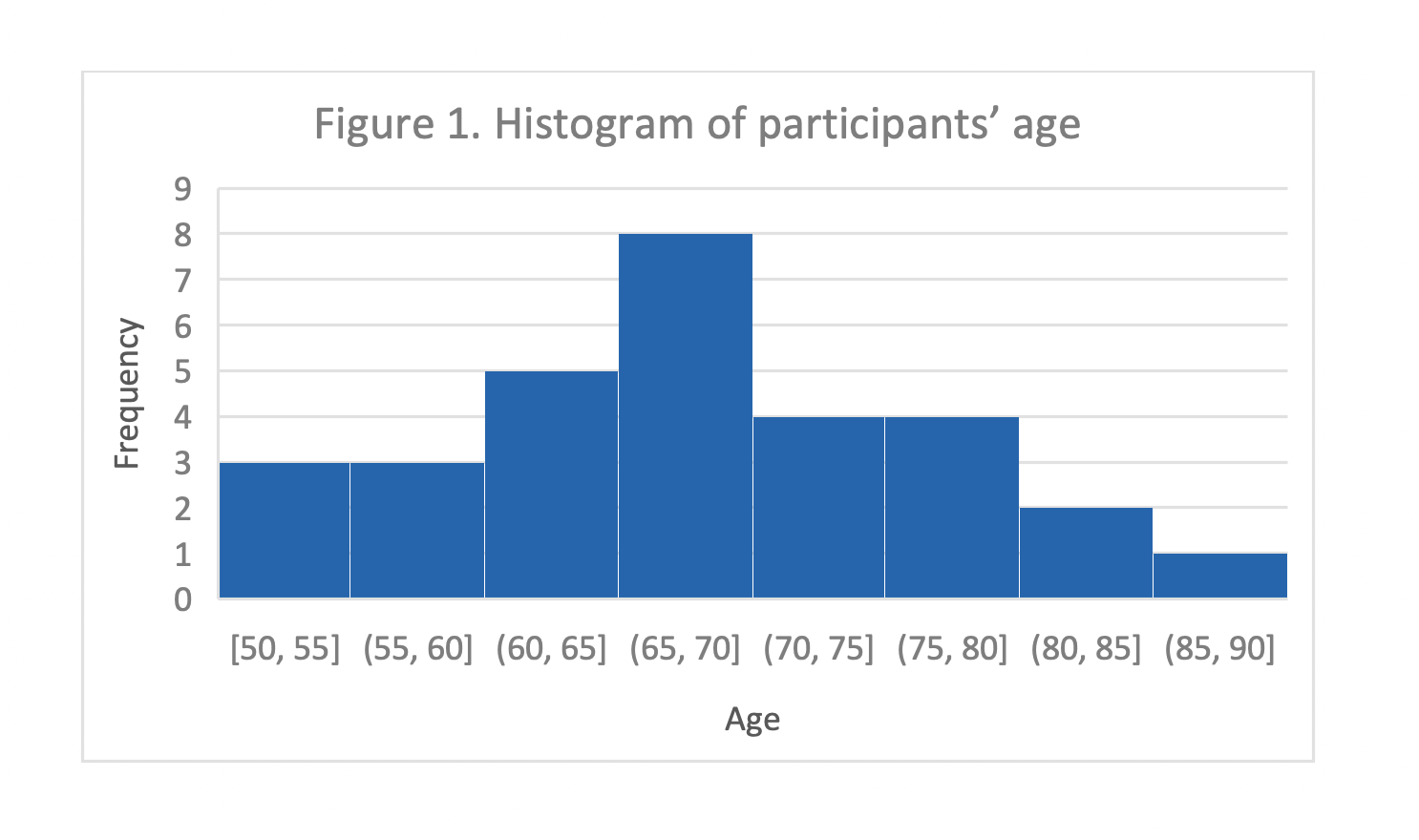
The sample of the FFallS Predictor investigational study (n= 30) consisted of 19 females (63.3%) and 11 males (36.7%), with a mean age of 68 years (9.3 SD). A detailed breakdown of ages can be seen in the histogram in Figure 1.
Further participant details can be observed in Table 1, with information on cardiovascular medical history, polypharmacy, BMI, frailty status, and fall risk, according to data gathered during the baseline assessments.
Study current status
• All 12-month follow ups are completed. Data collection is finished.
• 16 out of 30 (53.3%) participants have experienced falls; among these participants, 38 falls have occurred.
• Thanks to the Reveal LINQ ™, serious arrhythmias needing pacemaker implantations were detected in 5 participants, who have been subsequently withdrawn from the study.
• The study team is examining the data for possible patterns or relationships between changes in cardiac, physiological and gait variables and falls.
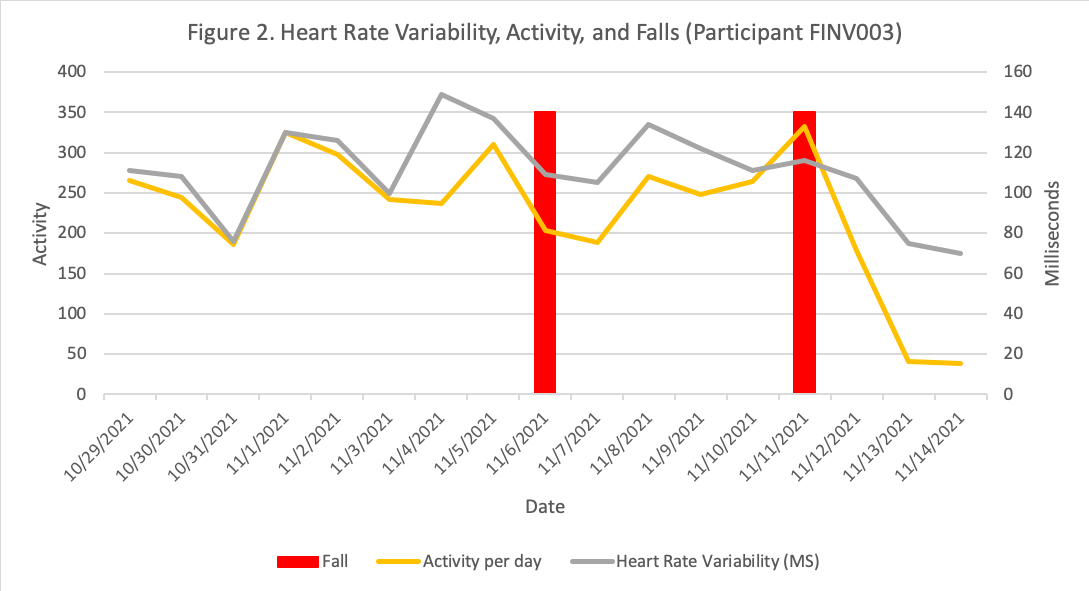
Example of data
Non-gait variables are also being explored, such as possible relationships between heart rate variability and activity before falls, as seen on example on Figure 2.